Explainer: What is a hydrogel?
Liquid water trapped in tangled polymers gives this material strangely useful properties

This close-up shows colorful hydrogel toy spheres known as “orbeez" or water beads. These are pretty when all plumped up but they're not meant for eating. If ingested they could continue to absorb water inside your body and cause dangerous blockages in the intestines.
Yulia Naumenko/iStock/Getty Images Plus
What can be made almost entirely of water, yet even at room temperature won’t be wet? A hydrogel. These water-based gels are among the most helpful materials you’ve likely never heard of.

Think of Jell-O and related sweet wiggly snack treats as forefathers of modern hydrogels. Those edible gelatins are also mostly water (about 90 percent in Jell-O’s case). But the water doesn’t leak out. That’s because thread-like molecules — called polymers — network throughout the hydrogel’s jiggly gelatin. Those polymers cling to water molecules like flies on a fly strip. The result is a strange substance that holds its shape (like a solid) yet retains some of liquid water’s life-sustaining properties.
If “you heat [Jell-O] up, it’ll actually liquefy,” notes Srinivasa Raghavan. He’s a biomolecular engineer at the University of Maryland in College Park. That ability to liquify sets edible gelatins apart from modern hydrogels, he says. Polymers in the edible ones temporarily stick to water, like hook-and-loop tape. Scientists classify that type as “physical” hydrogels. The newer types are known as “chemical” hydrogels. Their polymers are all linked permanently by chemical bonds.
Chemical hydrogels are especially important for making medical devices that must stay in contact with the body — or even remain inside it. Implants are one good example. Hydrogels find the body very hospitable because it, like them, is mostly water. (If you weigh 100 pounds, about 60 pounds of you is water. Most of that water is trapped, just as in a hydrogel. Our bodies tend to trap that water in our blood vessels and within the polymers that connect our cells.)
Here are some of the growing applications for today’s chemical hydrogels.
Lab-grown tissues. Imagine a burn victim who needs a skin transplant. Scientists can grow skin cells in petri dishes. But those cells would just develop into flat sheets. Lab-grown cells won’t form the organized layers found in our skin. That’s because cells in bodies grow on polymer scaffolds. Those scaffolds help liver cells grow into a liver shape. Similarly, they guide skin cells into layers. So today, many biologists supply human tissues grown in the lab with hydrogel frameworks. The same type of scaffolding is being used to make lab-grown steaks — ones that develop the meaty structure of a cow’s muscle.
Oxygen diffusers. The tear-moistened surface of your eye’s cornea allows oxygen to diffuse directly from the air into your eyeball. And that’s good. But when contact lenses cover the eyes, that can cut off exposure to much of that oxygen. To avoid that, soft lenses now depend on hydrogels. Their water-swollen polymers allow oxygen to reach the eye pretty much as normal.
Water absorbers. “We made a hydrogel that can absorb 3,000 times its weight in water!” Raghavan says. That, he thinks, “is a world record.” His team published the details of that study in Macromolecules back in 2014. Dried-out hydrogel beads slurp up water from their surroundings thanks to their water-loving polymers. It’s the same technology that allows virtually leak-proof disposable baby diapers. The U.S. Army even developed fancy, sweat-wicking underwear that traps moisture in a hydrogel.
Some growers also add dried hydrogel beads into pots of soil. When plants growing in the pots are watered, these beads soak up the moisture rather than letting it flow out the bottom or evaporate. This trapped moisture can then slowly diffuse back into the soil to slake the growing plants’ thirst in coming days.
Drug-delivery systems. Some medicines come packed in hydrogels. One such example is a pain-reliever for wounds and vascular disease known as Astero. It was designed to assist the healing of deep wounds by slowly releasing its contents into the moist tissues around the wound.
Impact protectors. In April 2022, Raghavan’s lab found that adding one new ingredient — cornstarch — gave hydrogels the ability to cushion fragile objects from breaking. You may have cornstarch in your kitchen. It’s often used to thicken a too-runny soup or pie filling. Raghavan’s Maryland team combined cornstarch with gelatin and plumped up the mix with water.
They jacketed some eggs in plain gelatin. Others were covered in the cornstarch-infused gel. Then they dropped each egg from a height of 30 centimeters (1 foot). Eggs covered in the plain-gelatin jackets smooshed into a mess upon landing. But those protected in starch-infused hydrogels landed intact every time.
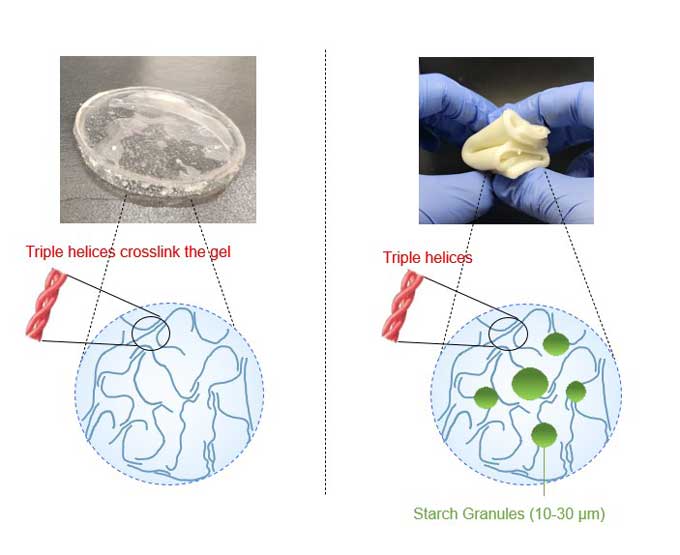
The neatest thing about this experiment, Raghavan says, is its simplicity. “I already had cornstarch in the lab,” he says. When a student suggested adding it to a hydrogel, a whole new application emerged.
One day, such a gel might be used to make a “case that protects your phone,” Raghavan says. Or it might protect an athlete’s head as better cushioning in a helmet. It might even find use as the basis of a new type of surgical implant. Each vertebra in the spine and each of our joints are naturally cushioned by little pillow-like discs of cartilage. When those discs get injured, surgeons repair or replace them with synthetic cartilage. These replacements do not contain water, Raghavan says. He thinks starch-enriched hydrogels might offer a more natural alternative.







